 Let us calculate the Van der Waals radius of Sb. Van der Waals radius is the closest distance between two atoms that are not attached through bonds. Let us see the boiling point of antimony.Īntimony possesses a very high boiling point of 1635 0 C or 2975 0 F. The boiling point is a particular temperature at which the liquid phase exists in equilibrium with the gas phase of that element. Let us find it.Īntimony has a relatively higher melting point of 630.63 0 C or 903.78 K. The melting point or liquefaction point is a definite temperature at which the solid and liquid state of an element exists in equilibrium. Let us calculate it for Sb.Īntimony has an atomic density of 6.691 g/cm 3. Antimony Atomic DensityĪtomic density depends on the atomic mass and the atomic volume of an element because atomic density is expressed as the ratio of the above two terms. But in Sanderson’s scale and Allred Rochow’s scale, the value of electronegativity is 2.46 and 1.82 respectively. Let us discuss it.Īntimony has electronegativity on the Pauling scale is 2.05. Antimony Electronegativity according to Paulingĭetermination of electronegativity of the element becomes significant to determine the bond polarity between the bond-forming atoms. The atomic weight of antimony is 121.76 amu (atomic mass unit). Antimony Atomic WeightĪtomic weight is determined by the total number of protons and neutrons along with consideration of the nuclear binding energy. Let us check the atomic numbers of Sb.Īntimony has an atomic number of 51 which indicates it has 51 protons in its nucleus. The atomic number is a unique property of an element because no two different elements have the same number of protons in their nucleus. 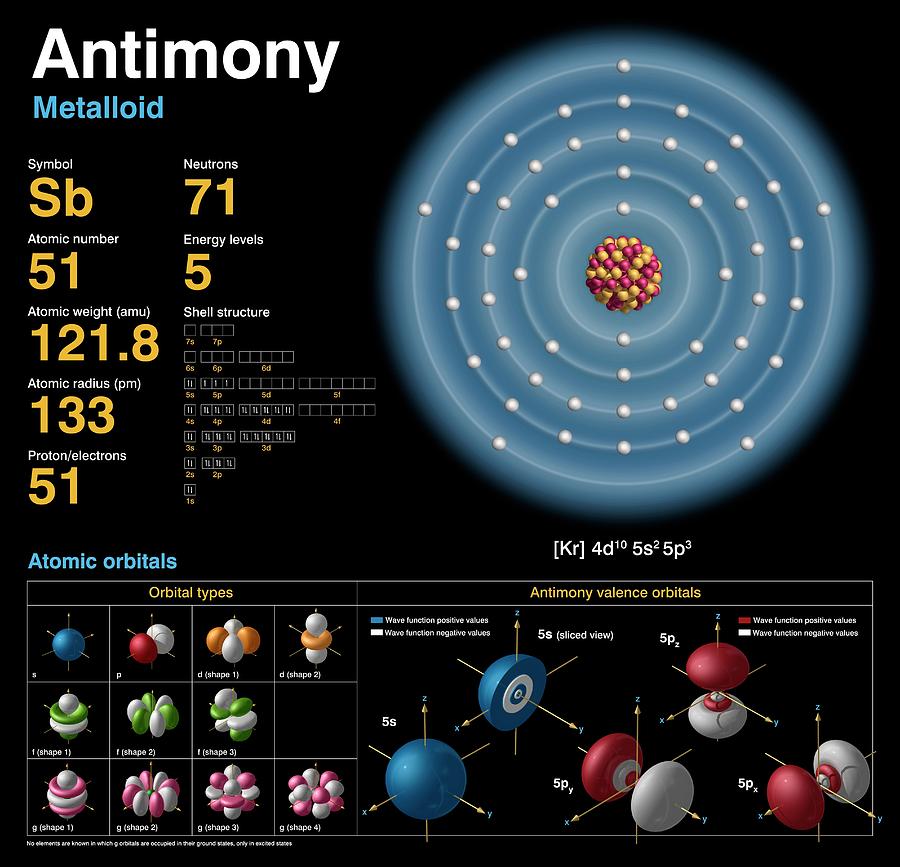
Let us see the clock of antimony.Īntimony belongs to the p-block because it has a partially filled p-block (5p 3) in its electron configuration. Antimony Block in Periodic TableĪ block in the periodic table consists of a set of elements having similar atomic orbitals, valence electrons, etc. The group number of antimony is 5 in the periodic table, and it is placed between tin (Sn) and tellurium (Te). The atoms placed in the same period (horizontal rows in the periodic table) show an equal number of electron shells in the outer part of the nucleus. It is placed between arsenic (As) and bismuth (Bi). 
Let us explore the group of Sb.Īntimony belongs to group 15 of the periodic table. The atoms belonging to the same group (vertical column in the periodic table) possess similar valence shell electronic configurations. This short form comes from the Latin name “Stibium”. Let us search for the symbol of antimony. Antimony SymbolĪ chemical symbol is a short form or abbreviation used to write or describe a chemical element. Let us describe the group and period of antimony in the periodic table, melting as well as boiling point, isotopes, allotropes, ionization energies, and chemical classification with detailed explanations. Elemental antimony possesses a layered structure that consists of a fused, ruffled six-membered ring. It is a hard, brittle metalloid compound. Let us focus on the chemical properties of antimony.Īntimony is refined industrially from stibnite by roasting and followed by reduction in presence of carbon or direct reduction of stibnite with iron. Antimony is a gray-colored lustrous metalloid that is found mainly as Sb 2S 3. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
